 Skin Relief Body Wash with Coconut Scent
Skin Relief Body Wash with Coconut Scent 
AVEENO® SAMPLES FOR YOUR PATIENTS
Receive news on the latest skincare innovations, sample opportunities, hot topics in literature, patient insights and resources for your patients.
Colloidal Oatmeal Therapeutic Scented Lotion and Wash Clinically Shown to Be As Well Tolerated and Gentle to Sensitive Skin As a Fragrance-Free Therapeutic Lotion and Creamy Wash
Judith Nebus1 , Heather Smith1 , Neena Tierney PhD1 , Joseph Fowler MD2 1 Johnson & Johnson Consumer Inc. Skillman, NJ, USA 2Dermatology Specialists, PSC, Louisville, KY, USA
ABSTRACT
- There is a wide range of body-moisturizing options available within the marketplace. Patients, including those with sensitive skin, tend to prefer the product experience of a scented formulation over that of a fragrance-free formulation. To minimize the potential risk of skin irritation and sensitization, dermatologists typically recommend fragrance free therapeutic products to their patients. With the ability to intensely moisturize, relieve irritation and help prevent the recurrence of extra-dry skin, it is important that therapeutic body moisturizers and cleansers are not only gentle and mild, but also have an aesthetic profile patients prefer to drive compliance and deliver on these benefits.
- In order to give people with sensitive skin better options when choosing therapeutic moisturizing and cleansing products, as well as help drive patient compliance, a colloidal oatmeal moisturizer and wash formulation was developed that used two novel scents (coconut and chamomile). The ingredients used to create these scents also provided skin conditioning benefits to the formulation. This study was conducted to evaluate the tolerance of both scented body wash and lotion regimens and compare them to a commercially available therapeutic fragrance-free regimen.
- This was a single-center, randomized crossover, controlled, blinded 4-week clinical use study. Eighty-nine subjects, aged 18 to 70 years, with history of self-perceived sensitive skin were enrolled. Subjects enrolled had previous documented skin sensitivities to household or skin care products. The study consisted of two 2×2 crossover sub-studies, one for each scented product regimen. The fragrance-free regimen consisted of a highly recommended therapeutic fragrance-free wash and lotion. At baseline (Visit 1), each subject was randomly assigned to one of the two cells (coconut or chamomile) and one of the two treatment sequences within the cells (scented versus fragrance free). This study consisted of five visits over a 4-week usage period. Visits were conducted at baseline and Weeks 1, 2, 3, and 4.
- The regimens were evaluated by a dermatologist via clinical tolerance assessments including rash, redness, burning, tightness, etc. Weekly self-assessment questionnaires were completed by the subjects. Statistical analysis of the clinical assessments found that both the coconut and chamomile colloidal oatmeal therapeutic scented wash and lotion regimens were as well tolerated as the non-scented regimen. Furthermore, self-assessments showed that the sensitive skin subjects found the scented regimens more pleasing to use when compared to the commercially available non-scented regimen. No product-related adverse skin reactions were observed with either of the scented regimens.
OBJECTIVES
- Compare the clinical tolerance of two scented colloidal oatmeal body wash and lotion regimens to a leading, commercially available fragrance free therapeutic body wash and lotion.
- Evaluate patient response to both scented and fragrance free regimens.
- Assess the coconut and chamomile colloidal oatmeal therapeutic scented regimens’ ability to provide beneficial therapeutic moisturization.
STUDY DESIGN
- Single-center, randomized crossover, controlled, blinded 4-week clinical use study.
- Eighty-nine subjects, aged 18 to 70 years, with reported sensitivities to common household/ skincare products.
- Two 2×2 crossover sub-studies, one for each scented product regimen.
- Clinical tolerance assessments evaluated at each of the 5 visits over a 4-week usage period and through a weekly self-assessment questionnaire.
RESULTS
Figure 1. Dermatologist Grading Tolerance Comparison between the mean grading scores for the studied clinical parameters after two weeks of using either the coconut or fragrance-free regimen. No statistically significant difference (P > 0.05) in tolerance between the coconut scented regimen and the fragrance-free regimen.
8.6_figure_1.jpg
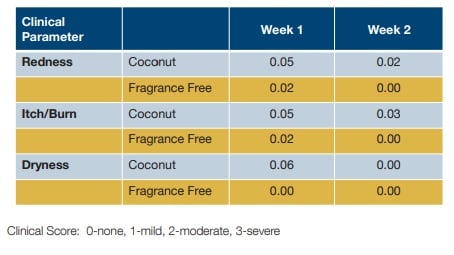
Figure 2. Self-Assessment Week 2
- The self-assessment mean scores after 2 weeks of treatment for both the chamomile and coconut regimens versus a leading fragrance free regimen:
- Scale 1-10, the higher the score, the more favorable the assessment
- Both scented regimens were comparable to the fragrance-free regimen for all parameters, and in some cases, outperformed the fragrance-free regimen.
8.6_figure_2.jpg
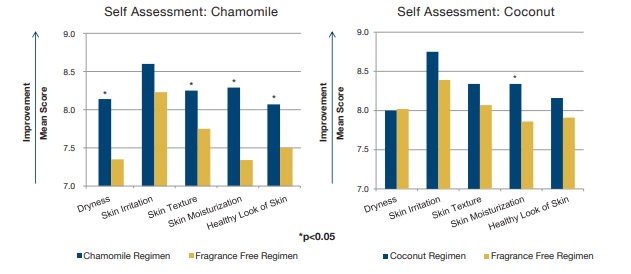
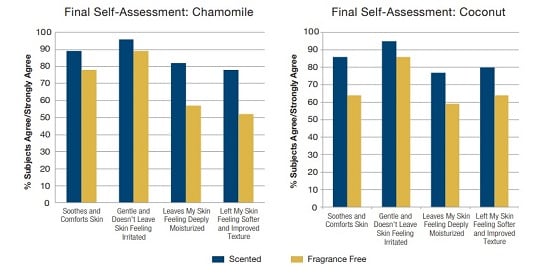
Figure 3. Final Self-Assessment
- Results of the final questionnaire given to subjects at the completion of each 4 week regimen Subject-perceived benefits to skin (after 2 weeks using the regimen)
- Subjects rated the scented regimen higher for comforting, soothing, and gentleness to the skin
8.6_figure_3.jpg
CONCLUSIONS
- Statistical analysis of the clinical assessments found that both the coconut and chamomile colloidal oatmeal scented regimens were as well tolerated as a leading fragrance free therapeutic regimen (P> 0.05).
- Self-assessments of various skin health parameters revealed that subjects found the chamomile and coconut colloidal oatmeal regimens as effective, if not more effective, than the fragrance-free regimen.
- Final self-assessments showed the subjects rated the gentle scented regimen higher for comforting, soothing, and gentleness to the skin when compared to a leading fragrance free therapeutic body wash and lotion regimen.
- Study demonstrated that the coconut and chamomile colloidal oatmeal therapeutic scented wash and lotion regimens provide beneficial therapeutic moisturization while maintaining a pleasant aesthetic.
- Final self-assessment questionnaires showed sensitive-skin subjects rated the scented colloidal oatmeal regimens higher for many aesthetic attributes when compared to the fragrance-free competitor, which can result in improved patient compliance.
ACKNOWLEDGEMENTS
- The authors wish to thank the participants in these studies. • Writing assistance was provided by Richard Bradley Rzendzian, PharmD, of Johnson & Johnson Consumer Inc. (Skillman, NJ, USA).
- Editorial assistance was provided by Alex Loeb of Evidence Scientific Solutions (Philadelphia, PA, USA), and was funded by Johnson & Johnson Consumer Inc.
CONFLICTS OF INTEREST
- This study was supported in full by Johnson & Johnson Consumer Inc. (Skillman, NJ, USA).
- Judith Nebus, Heather Smith, and Neena Tierney, PhD, are employees of Johnson & Johnson Consumer Inc.
- Joseph Fowler, MD, is a consultant to Johnson & Johnson Consumer Inc.



All Fields required, unless otherwise indicated
Will be used as your user name
By submitting your information above, you agree that the information you provide will be governed by our site's Privacy Policy.